Vining Lab at Penn Dental Medicine
and Penn Engineering
Bridging the gap between materials science, biology, and dental medicine to bring next-generation innovations to the clinic, Dr. Kyle Vining, DDS, PhD, practices restorative and cosmetic dentistry and leads a multi-disciplinary team of scientists and engineers investigating mechanical regulation of inflammation in cancer and regeneration. The Vining Lab values excellence, collaboration, and leadership. The Vining Lab’s overall goal is to discover new physical mechanisms of disease and to develop materials and therapies to radically disrupt the dental industry and ultimately transform oral, dental and craniofacial healthcare.
Current Areas of Investigation
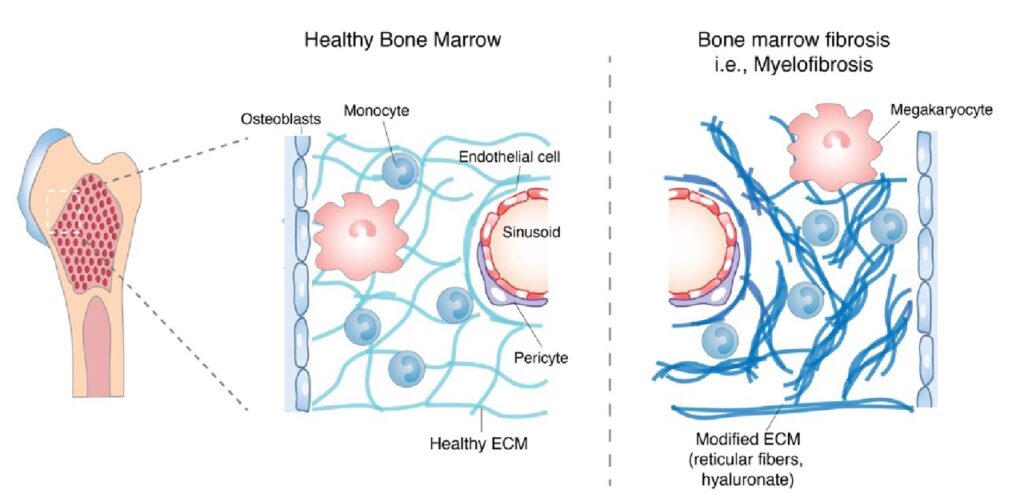 Fibrosis and remodeling of extracellular matrix (ECM) are involved in many diseases affecting health, such as tumors, wound healing, and chronic inflammation. During fibrosis, tissues undergo changes in their viscoelastic properties, i.e., how they resist deformation like a solid and dissipate stress over time like a fluid. Independent of stiffness, an applied stress relaxes rapidly in a more viscous, liquid-like matrix, whereas in a more elastic, solid-like material, stress relaxes slowly (Vining and Mooney, 2017). The Vining Lab investigates the impact of viscoelasticity on inflammation in fibrotic tissues and develops new immunotherapies in cancer.
Fibrosis and remodeling of extracellular matrix (ECM) are involved in many diseases affecting health, such as tumors, wound healing, and chronic inflammation. During fibrosis, tissues undergo changes in their viscoelastic properties, i.e., how they resist deformation like a solid and dissipate stress over time like a fluid. Independent of stiffness, an applied stress relaxes rapidly in a more viscous, liquid-like matrix, whereas in a more elastic, solid-like material, stress relaxes slowly (Vining and Mooney, 2017). The Vining Lab investigates the impact of viscoelasticity on inflammation in fibrotic tissues and develops new immunotherapies in cancer.
- We utilize an artificial ECM to dissect a mechanical checkpoint of monocyte fate and develop strategies to target myeloid inflammation in vivo. Monocytes are a type of white blood cells that impact the local immune response by producing cytokines and differentiating into phagocytic and antigen-presenting cells, like macrophages and dendritic cells.
- We first investigated this concept in hematopoietic malignancies that result in myelofibrosis (Vining, K.H., et al., Nature Materials, 2022). The data revealed changes of viscoelasticity in diseases associated with myelofibrosis, more than changes in stiffness alone, regulates inflammation and differentiation of monocytes, and is targetable by a PI3K-gamma inhibitor.
- This work has potential broad applications in fibrotic and inflammatory diseases.
Vining, K.H., Mooney, D.J. (2017). Mechanical forces direct stem cell behavior in development and regeneration. Nature Reviews Molecular Cell Biology 18, 728-742. PMCID: PMC5803560
Vining, K.H.*, Marneth, A.*, Adu-Berchie, K., Grolman, J., Tringides, C.M. , Liu, Y., Wong, W., Pozdnyakova, O., Severgnini, M., Stafford, A., Duda, G., Mullally, A., Hodi, F.S., Wucherpfennig, K., Mooney D.J., A mechanical checkpoint regulates monocyte differentiation in fibrotic niches. Nature Materials, July 2022. https://doi.org/10.1038/s41563-022-01293-3. *Authors contributed equally.
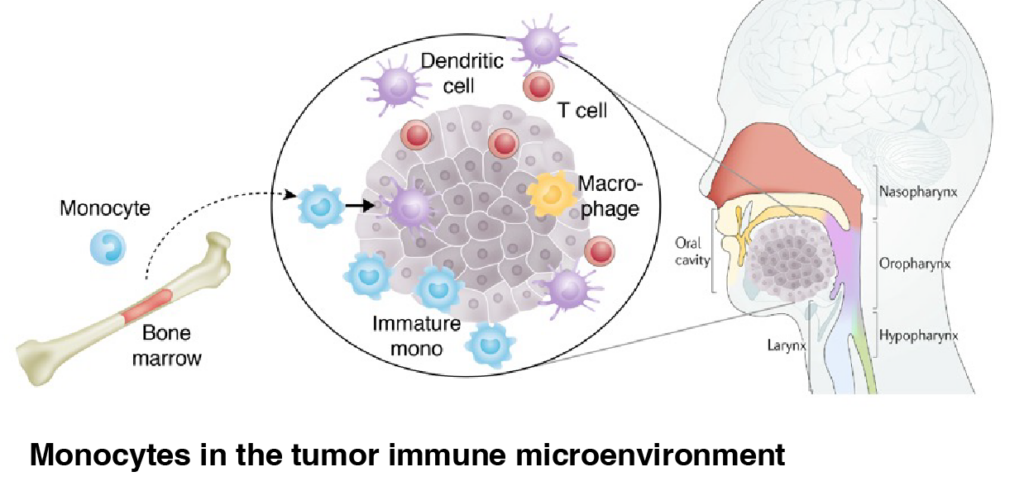 Head and neck squamous cell carcinoma (HNSCC) is the seventh most common cancer in the world and presents in most patients with locally advanced disease. There is an unmet clinical need to identify mechanisms of resistance in solid tumors, and to develop new strategies to boost the clinical response rate of immune checkpoint blockade.
Head and neck squamous cell carcinoma (HNSCC) is the seventh most common cancer in the world and presents in most patients with locally advanced disease. There is an unmet clinical need to identify mechanisms of resistance in solid tumors, and to develop new strategies to boost the clinical response rate of immune checkpoint blockade.
Monocytes are recruited to tumors and affect the immune microenvironment. Immature monocytes are largely considered immunosuppressive in nature, and support tumor evasion of anti-tumor immunity, whereas differentiated antigen-presenting cells, such as dendritic cells, are associated with improved clinical responses to immunotherapies. However, underlying factors that direct immature myeloid cells in tumors remain elusive.
The Vining Lab investigates whether mechanical cues regulate the fate of myeloid cells to alter the immune microenvironment in oral squamous cell carcinoma and fibrotic disease. Solid tumors are surrounded by a rigid stroma of ECM with collagen fibers that is more rigid than healthy tissue. In addition to stiffness, tumor stromal ECM exhibits relative fluid-like viscous and solid-like elastic properties, i.e., viscoelasticity.
- Vining earned a NIH K99/R00 Pathway to Independence Award from the NIDCR to investigate the relationship between mechanical regulation and monocyte fates in oral cancer through his post-doctoral position at Dana-Farber Cancer Institute in Boston, MA and starting his own lab at Penn Dental Medicine and Penn Engineering.
- We dissect mechanical regulation of myeloid cells to overcome immune resistance and boost efficacy of immunotherapies in head and neck cancer.
- We use a three-dimensional artificial ECM to study the impact of mechanical cues on differentiation human monocytes, analysis of clinical samples, and a mouse model of oral cancer.
- These studies have potential to lead to new treatments for HNSCC to over-come evasion of anti-tumor immunity.
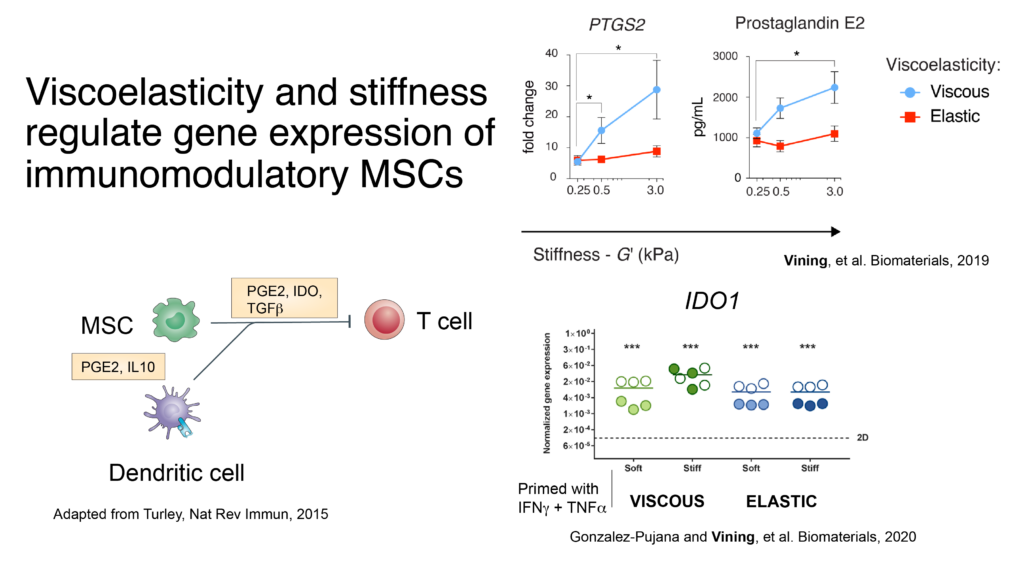 Our work investigates how mechanical properties of ECMdirects the immune fate of bone marrow niche cells. The immune system develops in the bone marrow, which is viscoelastic, exhibiting properties of both a solid and fluid. Dr. Vining earned a NIH K08 Mentored Clinical Scientist Career Development Award from the NIDCR to support his Ph.D. in David Mooney’s lab at Harvard University to develop artificial systems with human cells to study how the mechanical resistance of ECM can direct the immune fate of mesenchymal stromal cells (MSCs) (Vining, K.H., et al., 2018). An artificial fibrillar ECM was fabricated with interpenetrating networks of type-I collagen and chemically-modified polysaccharides. Viscoelasticity was specifically tuned independent of other material properties across a physiologic range of bone marrow stiffness. More fluid-like, viscous matrix was associated with immunomodulatory expression of MSCs, which is consistent with homeostasis in healthy bone marrow. Further, collaborative projects have demonstrated how programmable materials can be used to enhance persistence and immunomodulation of MSCs (Mao, A.S., et al., 2019), and to enhance licensing of MSCs for immunomodulation (Pujana-Gonzalez, A., Vining, K.H., et al., 2020).
Our work investigates how mechanical properties of ECMdirects the immune fate of bone marrow niche cells. The immune system develops in the bone marrow, which is viscoelastic, exhibiting properties of both a solid and fluid. Dr. Vining earned a NIH K08 Mentored Clinical Scientist Career Development Award from the NIDCR to support his Ph.D. in David Mooney’s lab at Harvard University to develop artificial systems with human cells to study how the mechanical resistance of ECM can direct the immune fate of mesenchymal stromal cells (MSCs) (Vining, K.H., et al., 2018). An artificial fibrillar ECM was fabricated with interpenetrating networks of type-I collagen and chemically-modified polysaccharides. Viscoelasticity was specifically tuned independent of other material properties across a physiologic range of bone marrow stiffness. More fluid-like, viscous matrix was associated with immunomodulatory expression of MSCs, which is consistent with homeostasis in healthy bone marrow. Further, collaborative projects have demonstrated how programmable materials can be used to enhance persistence and immunomodulation of MSCs (Mao, A.S., et al., 2019), and to enhance licensing of MSCs for immunomodulation (Pujana-Gonzalez, A., Vining, K.H., et al., 2020).
Vining, K.H., Stafford, A., Mooney, D.J. (2019) Sequential modes of crosslinking tune viscoelasticity of cell-instructive hydrogels. Biomaterials, 188, 187-197. PMCID: 30366219
Mao, A.S., Ozkale, B., Shah, N.J., Vining, K.H., Descombes, T., Zhang, L., Scadden, D.T., Weitz, D.A., Mooney, D.J. (2019) Programmable microencapsulation for enhanced mesenchymal stem cell persistence and immunomodulation. Proceedings of the National Academy of Science, 116 (31) 15392-15397. PMCID: PMC6681761
Gonzalez-Pujana A*, Vining K.H.*, Zhang DKY, Santos-Vizcaino E, Igartua M, Hernandez RM, Mooney DJ. Multifunctional biomimetic hydrogel systems to boost the immunomodulatory potential of mesenchymal stromal cells. Biomaterials. 2020 Oct;257:120266. PMCID: PMC7477339. *Authors contributed equally
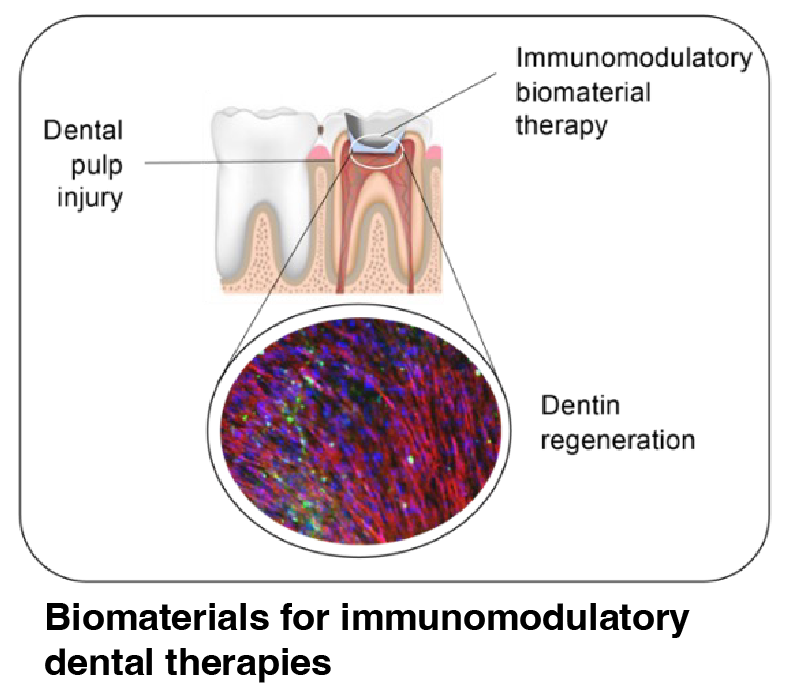 Untreated tooth decay in permanent teeth is the most common health condition worldwide, affecting over 3.5 billion patients according to the Global Burden of Disease 2017. Dental treatments directly cost $298 billion annually in 2010, with a total economic impact of an estimated $442 billion. There is an unmet clinical need to better understand how physical changes of ECM impact inflammation and how to promote repair and regeneration for clinical applications, such as dental pulpitis, periodontal disease, and orthodontic tooth movement. Dentists place hundreds of millions of dental fillings per year in the U.S. alone, which can result in dental pain or infection after the restoration is placed.
Untreated tooth decay in permanent teeth is the most common health condition worldwide, affecting over 3.5 billion patients according to the Global Burden of Disease 2017. Dental treatments directly cost $298 billion annually in 2010, with a total economic impact of an estimated $442 billion. There is an unmet clinical need to better understand how physical changes of ECM impact inflammation and how to promote repair and regeneration for clinical applications, such as dental pulpitis, periodontal disease, and orthodontic tooth movement. Dentists place hundreds of millions of dental fillings per year in the U.S. alone, which can result in dental pain or infection after the restoration is placed.
The Vining Lab aims to develop next-generation mechano- and biomaterial-based therapeutics that interface with oro-dental tissues for healing and regeneration (US Patent issued US11224679). This project will identify myeloid populations involved in dental pulp injury and will develop biomaterials to target pulpal inflammation.
- We hypothesize that irreversible dental pulp injury results in fibrosis of the extracellular matrix and increased pro-inflammatory myeloid cells.
- We correlate the mechanical properties of inflamed dental tissues by micro-rheology and atomic force microscopy with immune cell populations identified by flow cytometry, next-generation sequencing, and immuno-histochemistry.
- We develop biomaterials strategies to target dental pulp inflammation using experimental immuno-mechanical therapies, including a combination of chemical and immune checkpoint drugs and cellular therapies. Candidate therapies are tested in a model of dental pulp injury (Vining et al., Advanced Materials 2018)
- We also develop new artificial extracellular matrix hydrogels for regeneration of stem-cell derived dental tissues. Adult stem and progenitor cells are encapsulated in hydrogel with defined biochemical and mechanical cues to program develop and regeneration of craniofacial epithelial and mesenchymal tissues (Vining, Lombaert et al., Biomaterials, 2019).
- The goal of these studies is to develop new materials to drive dental regeneration by modulating immunomodulatory factors to enhance pulpal healing and dentinogenesis, and to explore through collaboration other applications in salivary glands, periodontal disease, and orthodontic tooth movement.
A.D. Celiz, K.H. Vining, D.J. Mooney. Methods and compositions for dental tissue repair and/or regeneration. US Patent 11,224,679. Issued 2022.
Vining, K.H., Scherba, J., Bever, A., Alexander, M., Celiz, A.D., Mooney, D.J. (2018) Synthetic Light-Curable Polymeric Materials Provide a Supportive Niche for Dental Pulp Stem Cells. Advanced Materials, 30, 1704486. PMCID: PMC5788014
Vining, K.H., Lombaert, I.M., et al. (2019). Neurturin-containing laminin matrices support innervated branching epithelium from adult epithelial salispheres. Biomaterials, 216, 119245. PMCID: PMC6720117




















